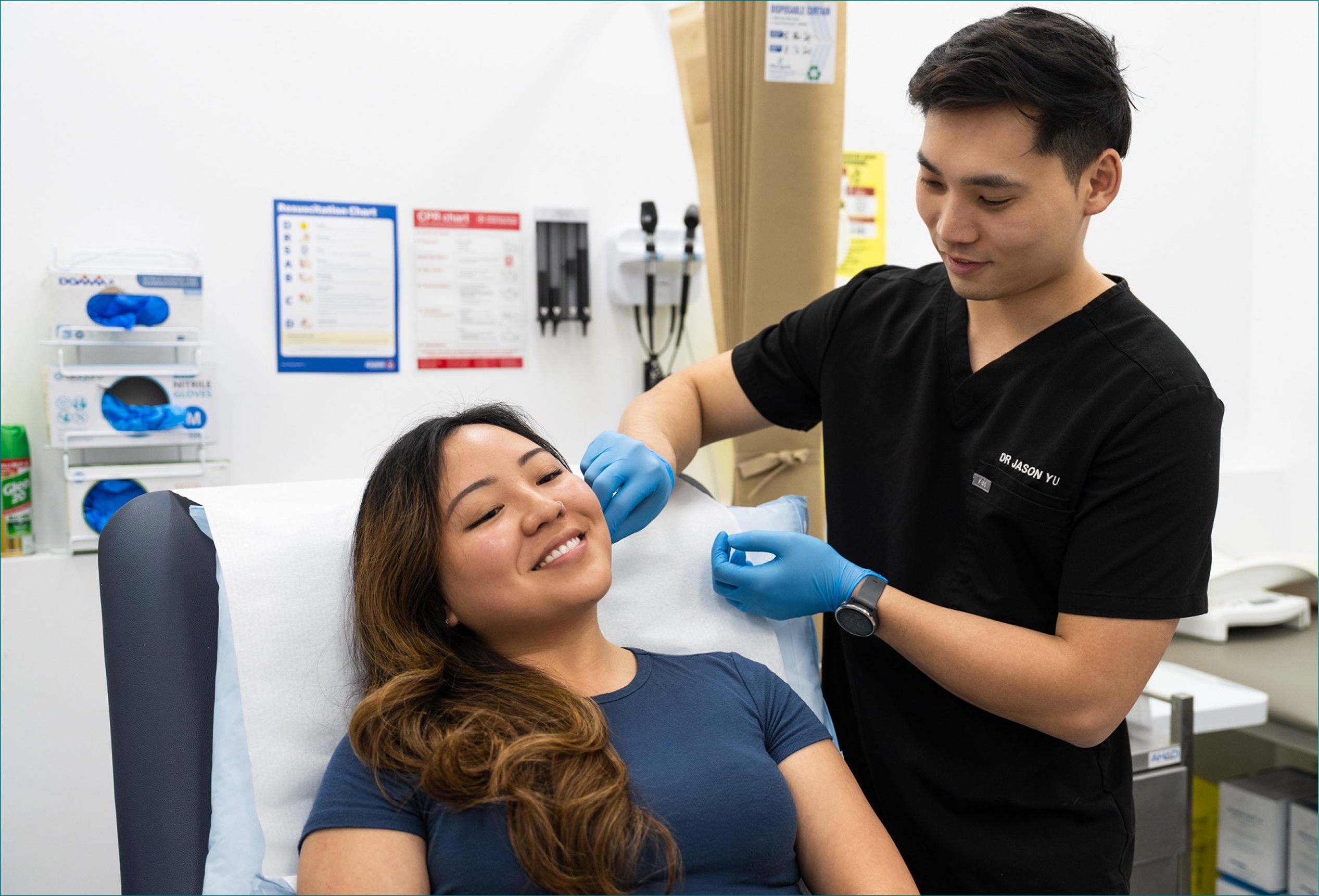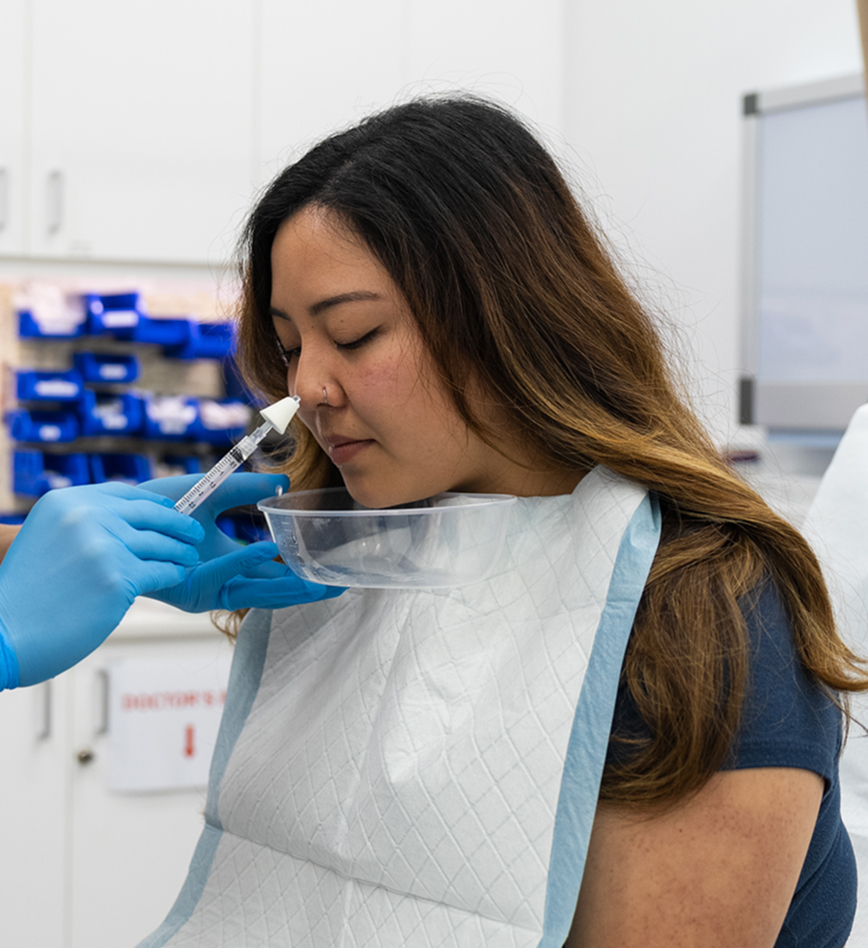
Formulated Nasal Atomisation
A specially formulated nasal mist delivered gently into the nasal passages to help relieve your allergy symptoms. Quick, easy and done within 2 minutes.
Formulated nasal atomisation delivers a specific medication directly to the nasal passages using a fine mist spray. The treatment works by blocking acetylcholine release from nerve endings in the nasal mucosa, helping to reduce mucous from the nose (1). Due to AHPRA advertising restrictions Dr Jason is unable to mention the specific medication being used.
This treatment may be considered for patients who experience persistent nasal discharge that has not responded adequately to conventionaltreatments such as antihistamines, or nasal sprays (2). Dr Jason will evaluate your medical history,symptoms, and previous treatment responses to determine if you are asuitable candidate.
Most patients find the procedure quite comfortable with minimal discomfort. The treatment involves delivering a fine mist into the nasal passages, which typically feels like a gentle nasal spray. There are no needles or injections involved in this method. You may experience mild, temporary feeling of nasal fullness or brief tingling, but any discomfort is usually very brief and well-tolerated (4).
Responsetimes vary between individuals. Some patients may notice improvements within hours to days of treatment, while others may require severalweeks to see the full benefit (5). Research studies show treatment effects lasting up to 3 months, with some patients experiencing benefits for 6 months (6,7).
Clinical studies demonstrate that the medication is generally safe and well-tolerated (8). Common temporary effects may include mild nasal irritation, temporary changes in nasal sensation, slight nasal congestion, or occasional mild nosebleeds immediately following treatment (9).
Medicare will cover part of the cost of the consultation with the doctor but does not cover this treatment. Health insurance companies do not cover this type of treatment either. Contact the clinic for a quote on the price.
The best way to determine if formulated nasal atomisation is appropriatefor your condition is through a comprehensive consultation with Dr Jason Yu. During this appointment, Dr Jason will review your medical history, examine your nasal passages, discussyour symptoms and previous treatments, and explain all available options. Together, you can make an informed decision about the most suitable treatment approach for your individual circumstances.
1. Botulinum Toxin forRhinitis. Ozcan C, Ismi O. 8, s.l. : Curr Allergy Asthma Rep, 2016,Current Allergy and Asthma Reports, Vol. 16.
2.The minimum effective dose of abobotulinum toxin A injection forallergic rhinitis: a dose-escalation randomized controlled trial.Piromchai P, Pornumnouy W, Saeseow P, Chainansamit S. 1, s.l. :Laryngoscope Investig Otolaryngol, 2021, Laryngoscope InvestigativeOtolaryngology, Vol. 6, pp. 6-12.
3.Minimally invasive application of botulinum toxin A in patients withidiopathic rhinitis. Rohrbach S, Laskawi R, Olthoff A, et al. 18,s.l. : Head Face Med, 2009, Head & Face Medicine, Vol. 5.
4.Effect of botulinum toxin type A on nasal symptoms in patients withallergic rhinitis: a double-blind, placebo-controlled clinical trial.Unal M, Sevim S, Doğu O, Vayisoğlu Y, Kanik A. 9, s.l. : ActaOtolaryngol, 2003, Acta Oto-Laryngologica, Vol. 123, pp. 1060-3.
5.Intranasal botulinum toxin type A for allergic rhinitis: arandomized, double-blind, placebo-controlled clinical trial.Shemshadi H, Azimian M, Onsori MA, Mazloum Farsi Baf M. 4, s.l. :Iran J Allergy Asthma Immunol, 2013, Iranian Journal of Allergy,Asthma and Immunology, Vol. 12, pp. 313-8.
6.Botulinum toxin for chronic rhinitis: A systematic review andmeta-analysis. Zheng M, Ruan G, Wu H, et al. 10, s.l. : Int ForumAllergy Rhinol, 2021, International Forum of Allergy & Rhinology,Vol. 11, pp. 1410-1419.
7.A comparison of the effects of botulinum toxin A and steroidinjection on nasal allergy. Yang TY, Jung YG, Kim YH, Jang TY. 3,s.l. : Otolaryngol Head Neck Surg, 2008, Otolaryngology–Head andNeck Surgery, Vol. 139, pp. 367-71.
8.Evaluation of the efficacy and safety of topical versus intraturbinalapplication of botulinum toxin type A for the treatment ofnoninfectious chronic rhinosinusitis. Kazem NG, Gaber MA, Mandour MF,Sheta AA. 1, s.l. : Pan Arab J Rhinol, 2022, Pan Arab Journal ofRhinology, Vol. 12, pp. 19-24.